Scientific Library: results
Publications & Application notes
Implementation and development of OCTET Red96e BLI platform
Download the posterInterlaboratory Evaluation of a User-Friendly Benchtop Mass Spectrometer for Multiple-Attribute Monitoring Studies of a Monoclonal Antibody
Download the posterNew 2D-LC–MS Approaches for the Analysis of In-Process Samples and for the Characterisation of mAbs in a Regulated Environment
Location
LCGC North America
Validation of a Monocyte Activation Test (MAT) using the PyroMAT™ kit
Download the posterConference

Is multi-attribute monitoring ready for QC? Insights from an interlaboratory study
During the 8th Bioproduction Congress organised by MabDesign, Arnaud Delobel presented the latest insi

2D-LC/MS in a regulated biopharma environment: challenges and applications
In this presentation, held during the 14th Multidimensional Chromatography Working, Arnaud Delobel covered the following topics:

Multiple-Attribute Monitoring in QC: feasibility and alternatives to the bottom up approach
During Waters European Biopharma Analytical Forum 2022 (Webaf), Claire Butré presented on Multiple-Attribute Monitoring in QC.
During her talk, she covered the following topics:

Bridging Maurice with iCE3 (icIEF) and PA800+ (CGE) systems
In this presentation, Géry Van Vyncht covered the following topics:

Use of Octet in a GMP environment: constraints and applications
Discover more about the use of Octet in a GMP regulated environment.
During this presentation, Arnaud Delobel goes through different subjects:
Webinar

Multi-Attribute Monitoring at Peptide or Subunit Levels
During this webinar, organised by Waters, Arnaud Delobel presents a method to demonstrate the effective implementation of Multiple Attribute Monitoring (MAM) in a regu

Multiple-Attribute Monitoring in QC: alternatives to the bottom-up approach
Webinar organised by Genovis

Characterisation of anti-HCP antibodies by 2D-Differential Gel Electrophoresis (2D-DIGE)
Host-cell proteins are a major class of process-related impurities in biotherapeutics that can impact the safety and efficacy of the product.

BioLayer Interferometry (BLI) technology as an alternative to Surface Plasmon Resonance (SPR) in biopharma regulated environment
Live event organised by SelectScience
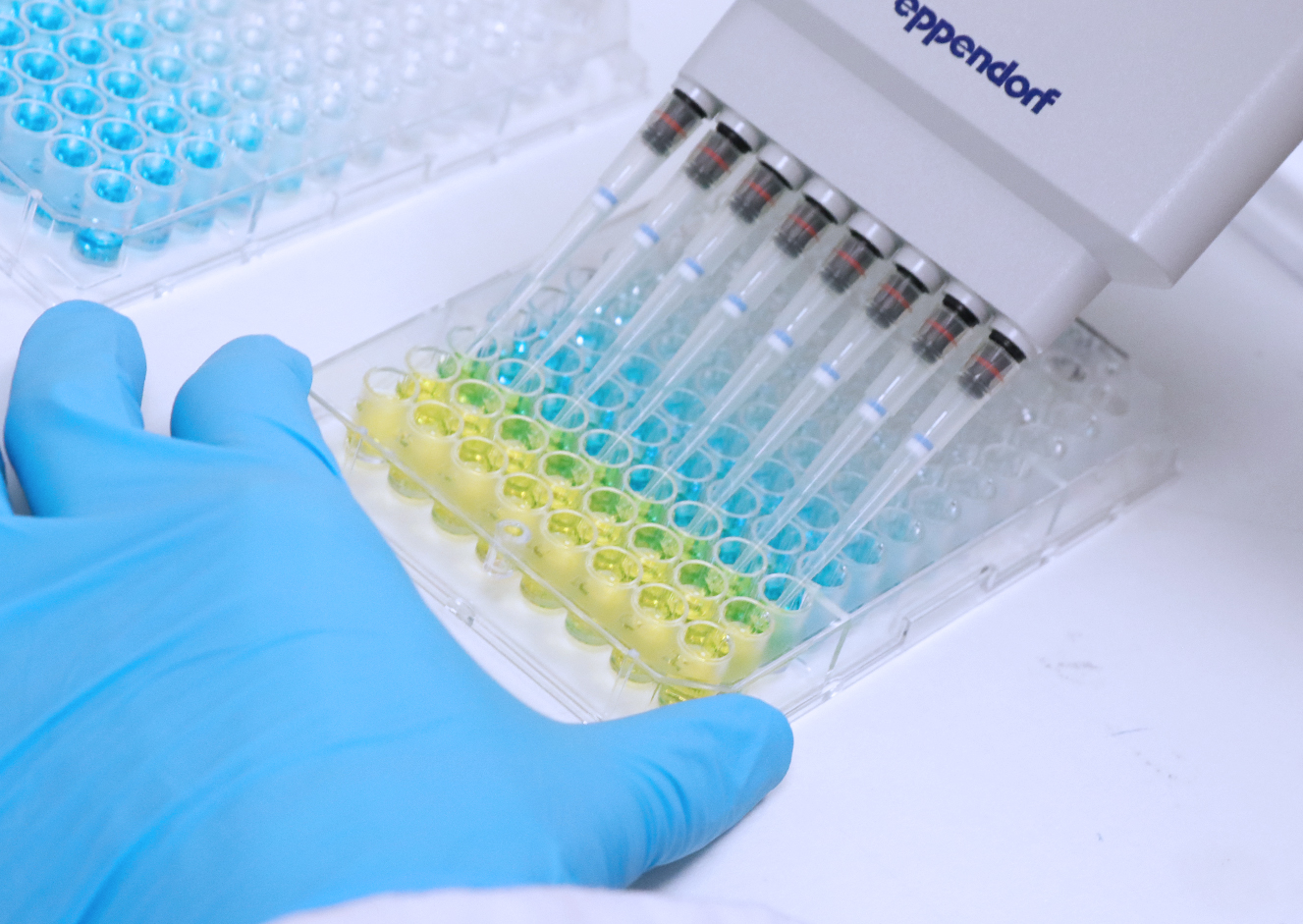
Pyrogens detection using Monocyte Activation Test (MAT)
Pyrogen testing is a regulatory requirement to ensure the product quality and safety of pharmaceutical products, as pyrogens can induce life threatening fever reactions. The reference method for endotoxin testing is the in vitro Limulus Amoebocyte Lysate (LAL) assay (Ph.
Glycan Analysis in Biopharma: challenges and future directions
Event organised by Separation Science, in collaboration with Agilent.
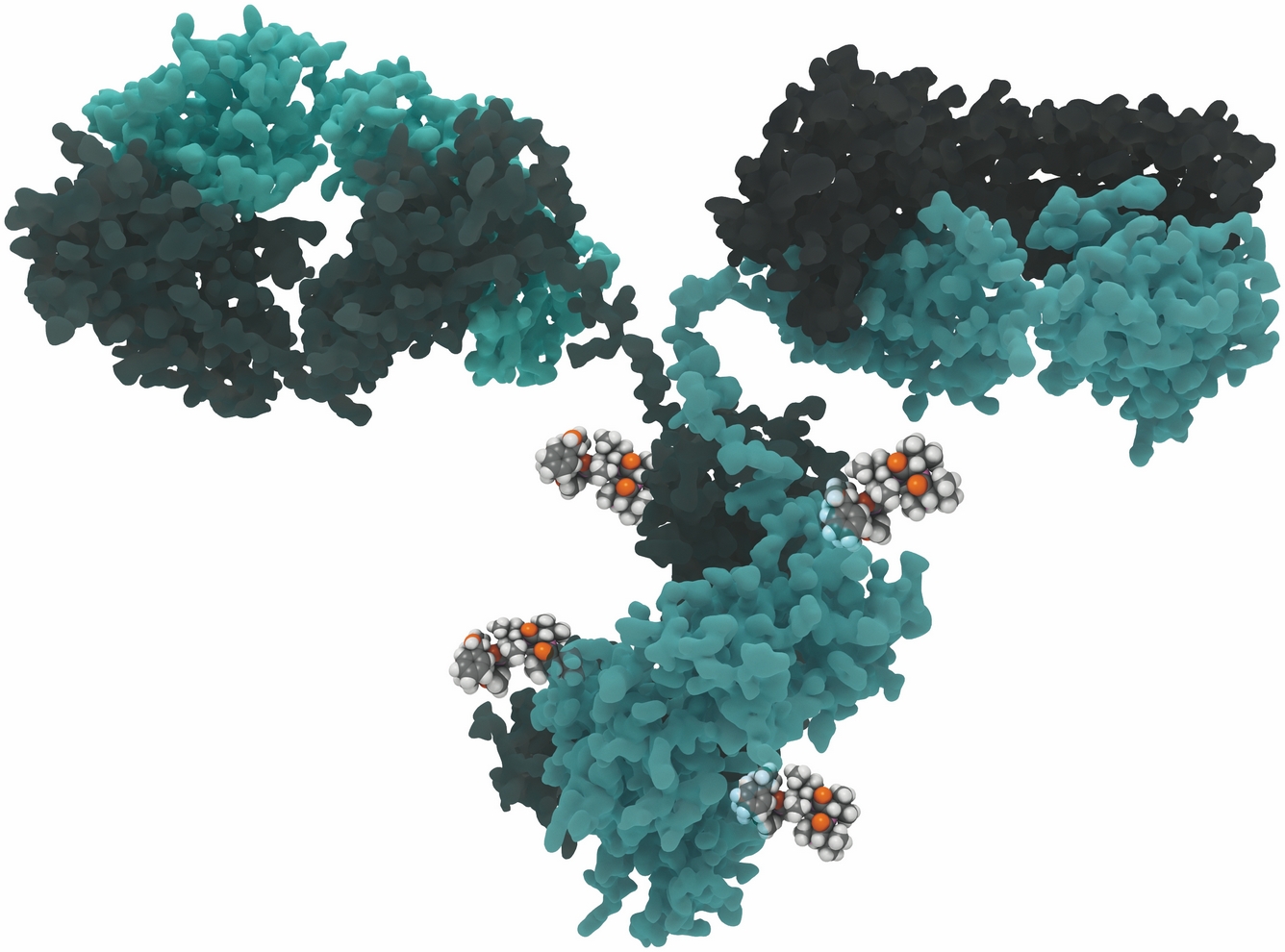
Development and validation of an antibody-drug conjugate bioassay
Antibody-drug conjugates (ADCs) are a class of drugs used in the treatment of different cancers. Unlike chemotherapy, ADCs are desi
Scientific poster
Interlaboratory evaluation of a user-friendly benchtop mass spectrometer for Multi-Attribute Monitoring studies
Download the posterA novel trypsin for accurate characterisation of biotherapeutic proteins with peptide mapping
Location
ASMS Conference on Mass Spectrometry and Allied Topics